IB Chemistry S2.2 Notes
This page contains our IB Chemistry notes for S2.2. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Covalent bonding & forces'.
Chapters
Covalent bonds
The next intramolecular bond to learn is the covalent bond. This is defined as the electrostatic attraction between a pair of nuclei and their shared pair of electrons. This is the strongest type of intramolecular bond and typically occurs between non-metals.
During a single covalent bond, two electron orbitals with one electron each overlap so that the electrons are paired in what is known as a molecular orbital. The remaining pairs of electrons are left in the valence shell of the atoms involved and become known as “lone pairs”.
There are three types of covalent bonds:
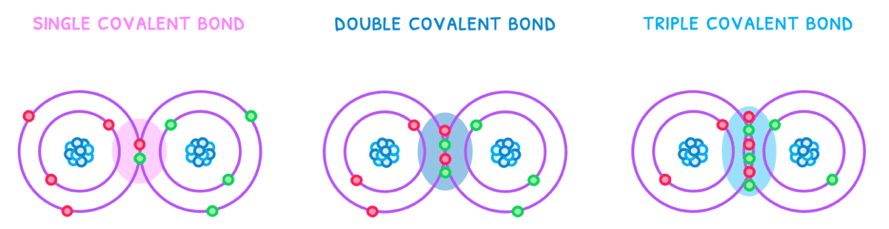
- Single - one pair of electrons is shared. This is therefore the weakest and longest bond.
- Double - two pairs of electrons are shared. This is therefore the bond of intermediate strength and length.
- Triple - three pairs of electrons are shared. This is therefore the strongest and shortest bond.
This is summarized in the table below:
| Bond type | Bond strength | Bond length |
|---|---|---|
| single | weakest | longest |
| double | intermediate | intermediate |
| triple | strongest | shortest |
tibertutor.com
Next Up
You have completed the sub-topic S2.2 notes, covering "Covalent bonding & forces" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic S2.2 resources