IB Chemistry S2.2 Notes
This page contains our IB Chemistry notes for S2.2. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Further covalent bonding'.
Chapters
Loading progress...
Further VSEPR
You need to know the additional two domains:
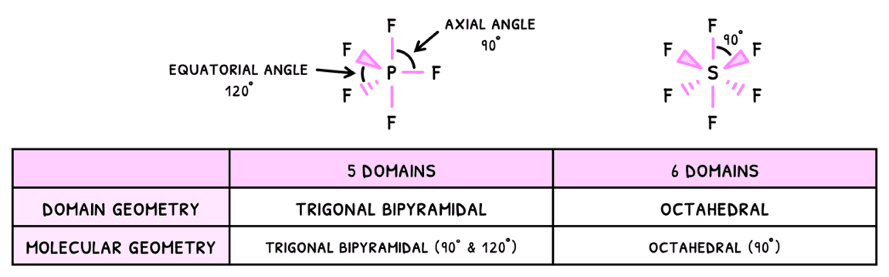
- Trigonal bipyramidal - 5 electron domains
- Octahedral - 6 electron domains
Just as before, the presence of a lone pair will decrease the bond angles by 2.5°, and this effect compounds. However, in some cases the pairs cancel one another and so the bond angles do not decrease.
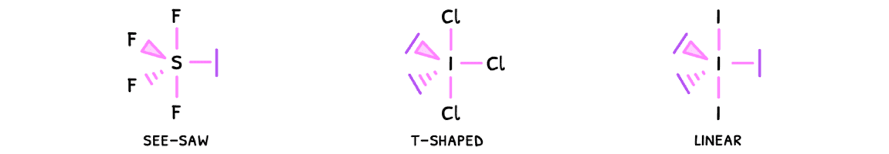
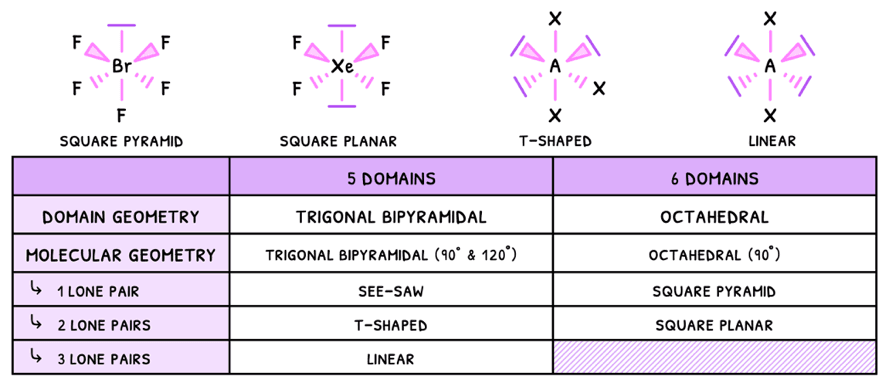
- Linear - electrons are removed from the equatorial plane, so the angle here becomes 180°.
- Square planar - electrons are removed from the axial plane, so the angles become 90°.
tibertutor.com
Next Up
You have completed the sub-topic S2.2 notes, covering "Further covalent bonding" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic S2.2 resources