IB Chemistry S3.1 Notes
This page contains our IB Chemistry notes for S3.1. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Periodic table & trends'.
Chapters
The periodic table
Dmitri Mendeleev is credited with creation of the first periodic table, which was generally accepted. With minor changes, this became the modern periodic table. In IB chemistry, having a good grasp of the periodic table will help you answer questions quickly and efficiently.
To start, it is important to understand that the periodic table is a table of all known elements. Since every element is distinguished from another by their atomic number (number of protons), they are arranged in order of increasing atomic number.
Then, each row is called a period and each column is called a group. The number of elements in each period and group is dictated by the electron configuration.
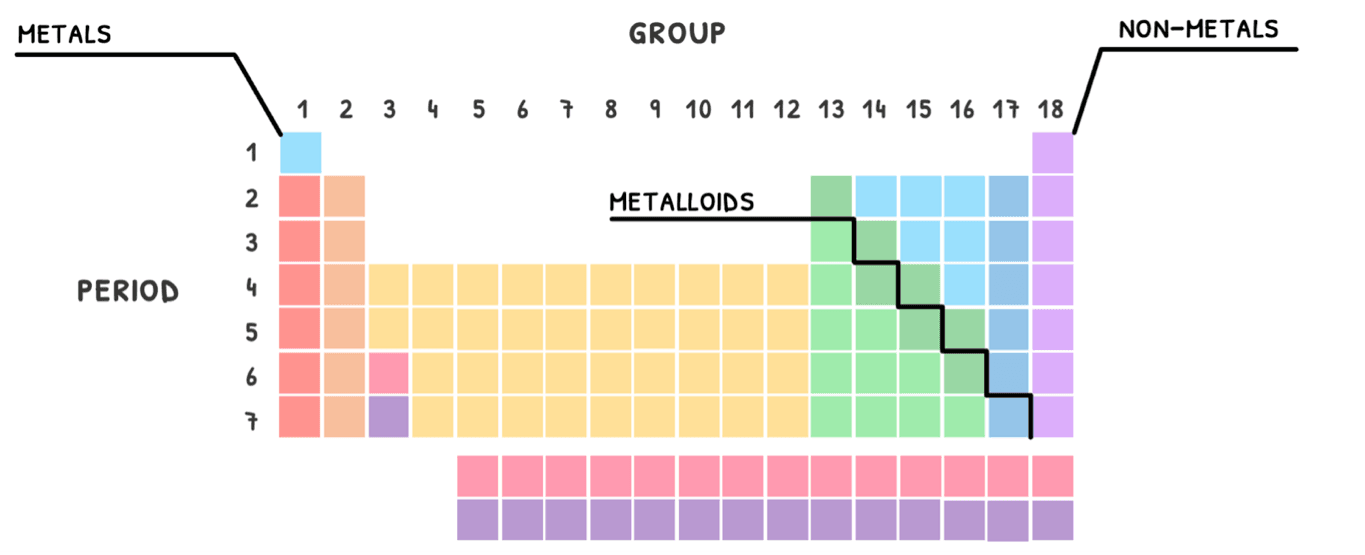
- The period indicates the number of energy levels the element has electrons in.
- The group indicates the number of electrons in the outermost energy level, called valence electrons.
Additionally, this organization also group elements by their properties. As a rule of thumb, the left and middle of the table is classed as metals, the right as non-metals, and in between a line of metalloids.
tibertutor.com
Next Up
You have completed the sub-topic S3.1 notes, covering "Periodic table & trends" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic S3.1 resources