IB Chemistry Topic 4 Notes
This page contains our IB Chemistry notes for topic 4. By reading each one of these notes, you will fully cover the content for IB Chemistry 'What Drives Chemical Reactions'.
Chapters
R1.1: Enthalpy
R1.2: Cycles of energy
R1.2: Further energy cycles (HL)
R1.3: Fuels
R1.4: Entropy & Spontaneity (HL)
Heat
In Topic S1.1, you learned that bond formation and breaking requires or releases energy. In this topic, you learn how to understand why this occurs and calculate the associated energy changes, particularly in relation to the heating of substances.
Remember that temperature is the average kinetic energy of particles. From this, you can appreciate that when fast particles from hot objects react with slow particles from cold objects, they exchange energy. Heat (Q) is thus the thermal energy transferred between two objects with different temperatures.
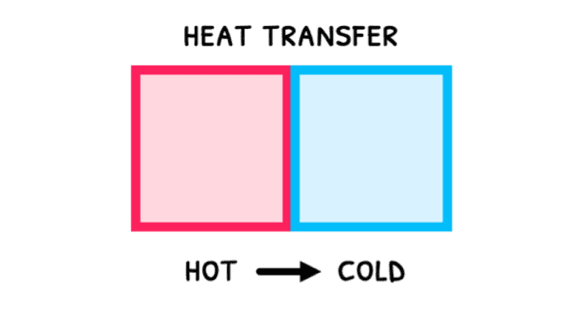
Since the faster particles tend to give up energy during collisions, heat naturally flows from hot objects to cold objects, or high temperatures to low temperatures. During this:
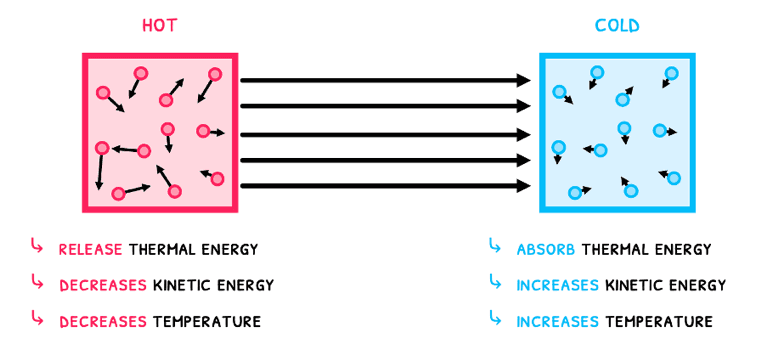
- Hot objects release thermal energy, which decreases the kinetic energy of their particles. As a result, their temperature decreases.
- Cold objects absorb thermal energy, which increases the kinetic energy of their particles. As a result, their temperature increases.
tibertutor.com
Next Up
You have completed the topic 4 notes for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other topic 4 resources