IB Physics Sub-topic B3 Notes
Moles & Molar Mass
In Topic B.1, you learned about phases, phase changes, and heat transfer of substances. Whilst it is easy to perform experiments measuring these phenomena in solids and liquids due to their finite volume, it completely changes in gases.
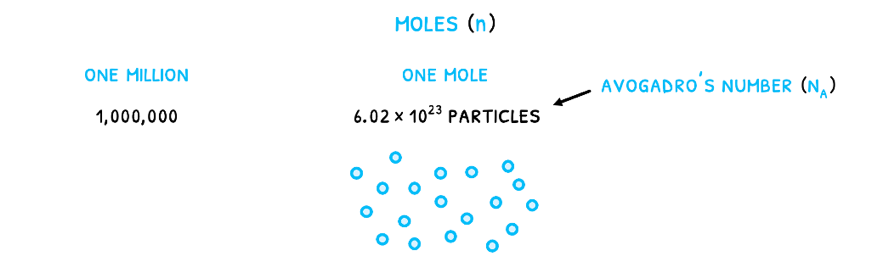
The change is that gases cannot easily be measured by their mass or volume. As a result, they are measured in moles (n). This is a measure of the number of particles given by Avogadro’s number (NA) equal to 6.022 x 1023 particles/mole. It relates to the number of particles that would make Carbon-12 exactly 12 grams as the standard element. Therefore, just like one million is 1,000,000, one mole is 6.022 x 1023 particles. For any number of particles (N), to calculate the number of moles, use the formula:
Note that this describes the number of particles so 1 mole of any diatomic molecule, such as H2, has 1.2044 x 1024 atoms because every molecule (and therefore particle) has two component atoms.
The mass of an atom can be expressed as molar mass (M), the mass of one mole of a substance in gmol-1. Calculating molar mass is possible with the formula: