IB Chemistry Sub-topic S1.4 Notes
This page contains our IB Chemistry notes for sub-topic S1.4. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Moles'.
Chapters
Loading progress...
Moles
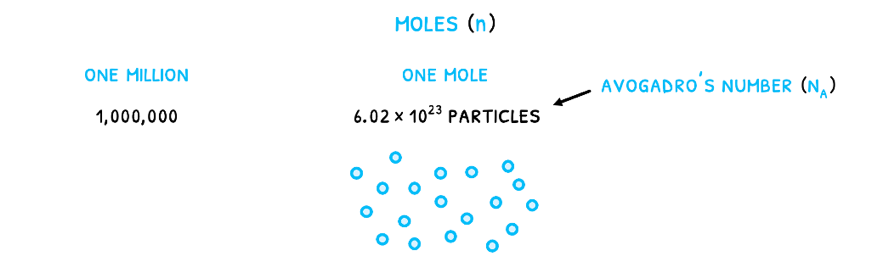
In reactions, substances are often measured in moles (n). This is a measure of the number of particles given by Avogadro’s number (NA) equal to 6.022 x 1023 particles/mole. It relates to the number of particles that would make Carbon-12 exactly 12 grams as the standard element. Therefore, just like one million is 1,000,000, one mole is 6.022 x 1023 particles.
Note that this describes the number of particles so 1 mole of any diatomic molecule, such as H2, has 1.2044 x 1024 atoms because every molecule (and therefore particle) has two component atoms.
tibertutor.com
Great Work!
You have now covered all of our sub-topic S1.4 notes, covering "Moles" for IB chemistry.
Now that you have completed these IB chemistry sub-topic S1.4 notes, covering "Moles", dive into our sub-topic S1.4 flashcards for the IB chemistry course.
IB chemistry sub-topic S1.4 flashcards
Explored IB Chemistry?
Get stuck into one of our other subjects!
Join 85,000 students, across 130+ countries, in 500+ IB schools. That's half of the IB science graduates worldwide.
Start a 7d free trial