IB Chemistry S1.2 Notes
This page contains our IB Chemistry notes for S1.2. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Further atoms'.
Chapters
Mass spec intro
Lastly, awkwardly nestled in this subtopic is the idea of mass spectrometry. In this subtopic, you need to understand that a mass spectrometer is a device used to:
- Determine the fragments of a compound, covered in more detail in Topic S3.2 HL.
- Determine an element's relative atomic mass from its isotopic composition.
In both scenarios, the working principle is the same, but the produced information is used differently. To determine fragments, the highest peaks are used. However, you will notice that several peaks exist for each fragment, and particulary so at the M+ peak. Let's explain this:
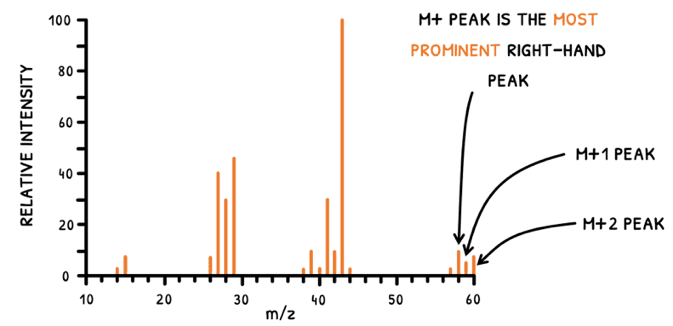
- An element may have two isomers with different atomic masses.
- A mass spectrometer analyzes multiple molecules within a sample, averaging their fragment masses to produce the mass spectrum.
- If a sample contains two isomers with:
- A mass difference of 1, the M+ and M+1 peaks are seen.
- A mass difference of 2, the M+ and M+2 peaks are seen.
There are three main isotopes to learn to easily spot these peaks:
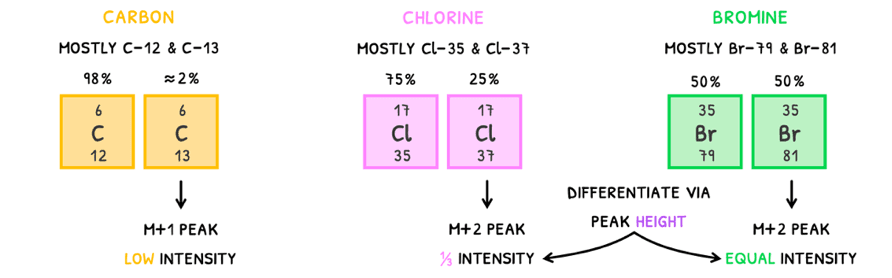
- Carbon - mainly exists as Carbon-12 (98%) and Carbon-13 (≈2%). The Carbon-13 isomer will produce an M+1 peak but because of its low abundance, the peak will have low intensity.
- Chlorine - mainly exists as Chlorine-35 (75%) and Chlorine-37 (25%). The Chlorine-37 isomer will produce an M+2 peak and because of its abundance, the peak will have an intensity of 33% relative to the M+ peak.
- Bromine - mainly exists as Bromine-79 (50%) and Bromine-81 (50%). The Bromine-81 isomer will produce an M+2 peak and because of its abundance, the peak will have an intensity equal to the M+ peak.
tibertutor.com
Next Up
You have completed the sub-topic S1.2 notes, covering "Further atoms" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic S1.2 resources