IB Chemistry S1.2 Notes
This page contains our IB Chemistry notes for S1.2. By reading each one of these notes, you will fully cover the content for IB Chemistry 'The atom'.
Atoms
In Topic S1.2, the primary focus is understanding atoms and the behavior of their electrons. This is very important because all chemical reactions will involve interactions between atoms and their electrons.
Atoms are defined as the smallest unit of an element. The IB requires you to know the Bohr model of the atom, which is composed of:
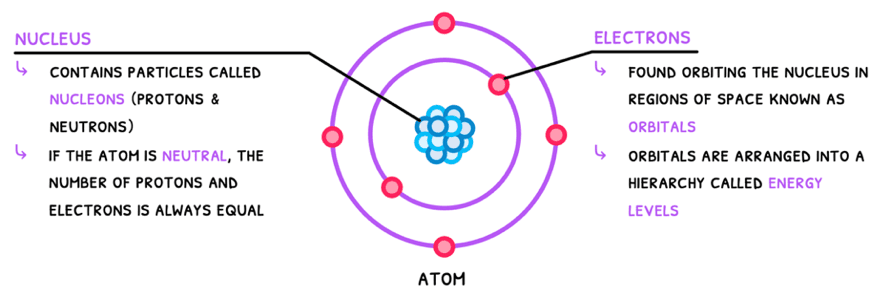
- A dense positively charged nucleus with protons and neutrons, termed nucleons.
- Negatively charged electrons in orbitals around the nucleus, arranged into energy levels/shells.
Additionally, you need to be aware of the basic properties of each component of the atom. This is succinctly summarized in the table below:
| Proton | Neutron | Electron | |
|---|---|---|---|
| Relative mass | 1 | 1 | 0 |
| Relative charge | +1 | 0 | -1 |
Although the Bohr model is technically incorrect, as electrons do not orbit the nucleus like satellites, it clearly communicates the concept of electrons having different energy levels. This is explored in more detail in Topic S1.3.
tibertutor.com
Next Up
You have completed the sub-topic S1.2 notes, covering "The atom" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic S1.2 resources