IB Chemistry S1.3 Notes
This page contains our IB Chemistry notes for S1.3. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Electron configurations'.
Chapters
Electromagnetic spectrum
Whilst you know about electrons and their orbitals in Topic S1.2, you also need to understand the discovery electron energy levels and transitions between them. This is evidenced by the absorption and release of energy by electrons in the form of an electromagnetic wave.
Electromagnetic waves are also known as electromagnetic radiation, which are a form of energy that travel as a wave. Remember that the basic components of waves are velocity (c), wavelength (λ), and frequency (f).
Whilst all electromagnetic radiation has a velocity of 3 x 108 ms-1, wavelength (λ), and frequency (f) change to give different wave types and energies. These are all described by the electromagnetic spectrum, wherein:
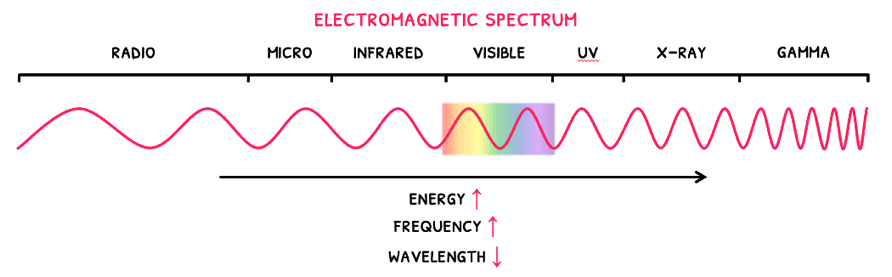
- As wavelength decreases, frequency increases.
- As wavelength decreases, energy increases.
Since the electromagnetic spectrum shows all the wave types, it is known as a continuous line spectrum. This is more specifically defined as a spectrum that shows all possible wavelengths. On the contrary, a line spectrum only shows particular wavelengths.
tibertutor.com
Next Up
You have completed the sub-topic S1.3 notes, covering "Electron configurations" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic S1.3 resources