IB Chemistry R1.2 Notes
This page contains our IB Chemistry notes for R1.2. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Cycles of energy'.
Hess's law
In Topic R1.1, you learned about enthalpy change and how to measure it for exothermic reactions using a calorimeter. Whilst there are other ways to directly measure enthalpy changes, it is not always possible for every reaction. For this, another method is needed.
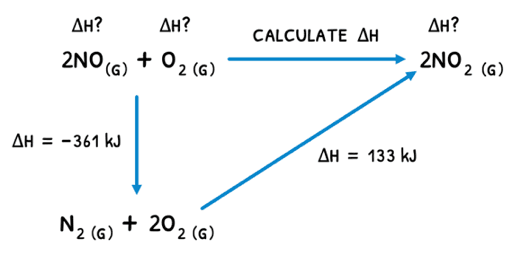
Hess's law describes that the enthalpy changes of a reaction is independent of the pathway between the initial and final states. This means that:
- From A to C, an enthalpy change of 10 kJ mol-1 may be observed.
- From A to B to C, the same end result was achieved and thus the overall enthalpy change is 10 kJ mol-1.
By breaking down a reaction into two sub-reactions, a reaction triangle can be formed and the unknown enthalpy change for the target reaction calculated.
tibertutor.com
Next Up
You have completed the sub-topic R1.2 notes, covering "Cycles of energy" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic R1.2 resources