IB Chemistry R2.3 Notes
This page contains our IB Chemistry notes for R2.3. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Further extent of change'.
The Reaction quotient
Now you can see how the equilibrium constant is the ratio of rate expressions. As a result, the formula only applies when the system is at equilibrium. If it has not reached this point yet, it is still in the reaction and the same formula describes the reaction quotient (Q). So in the reaction:
aW + bX ⇌ cY + dZ
The formula for this is:
The reaction quotient describes where the reaction lies with respect to the equilibrium.
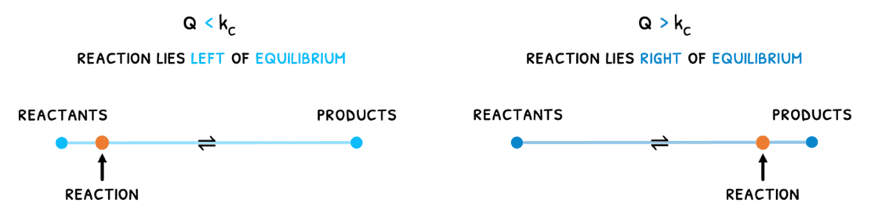
- If Q < Kc, it means the reaction still has more reactants and is undergoing a forwards reaction to produce more products and reach equilibrium.
- If Q > Kc, it means the reaction still has more products and is undergoing a reverse reaction to produce more reactants and reach equilibrium.
Lastly, if Q = Kc, it means equilibrium has been reached.
tibertutor.com
Next Up
You have completed the sub-topic R2.3 notes, covering "Further extent of change" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic R2.3 resources