IB Chemistry R2.2 Notes
This page contains our IB Chemistry notes for R2.2. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Rate of change'.
Chapters
Rate of Reactions
Now that you understand atoms, molecular structure, and how energy changes during reactions, you can focus on the reactions themselves. In Topic R2.2, you start this by focusing on the rate of reaction. This is simply the speed at which a reaction occurs and can be thought in a few ways:
- The increase of concentration/mass/volume of products over time.
- The decrease of concentration/mass/volume of reactants over time.
As a result, the units of rate can be mol dm-3 s-1, g s-1 or dm3 s-1. However, mol dm-3 s-1 is the standard unit of measurement for rate.
This is typically represented graphically with volume/mass/concentration on the y-axis and time on the x-axis. In these:
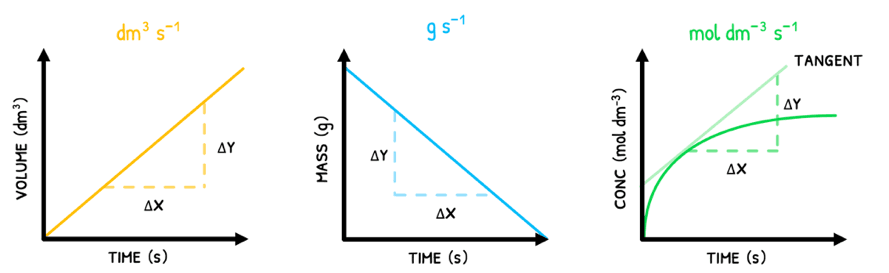
- In straight-line graphs, the rate at any time point is equal to the slope of the line.
- In curved-line graphs, the rate at any time point is equal to the slope of the tangent.
Note that products will show a positive slope because they are produced throughout the reaction whereas reactants will show a negative slope because they are used up throughout the reaction.
tibertutor.com
Next Up
You have completed the sub-topic R2.2 notes, covering "Rate of change" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic R2.2 resources