IB Chemistry S1.4 Notes
This page contains our IB Chemistry notes for S1.4. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Moles'.
Moles
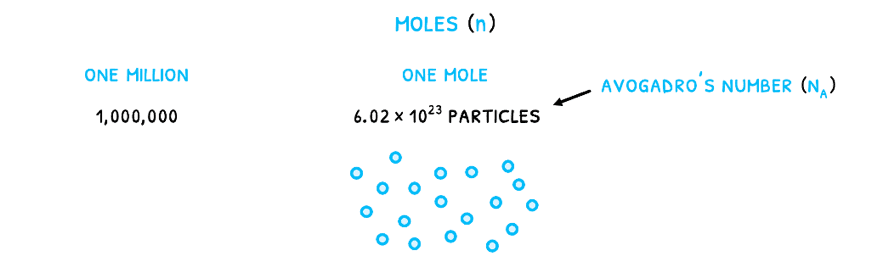
In reactions, substances are often measured in moles (n). This is a measure of the number of particles given by Avogadro’s number (NA) equal to 6.022 x 1023 particles/mole. It relates to the number of particles that would make Carbon-12 exactly 12 grams as the standard element. Therefore, just like one million is 1,000,000, one mole is 6.022 x 1023 particles.
Note that this describes the number of particles so 1 mole of any diatomic molecule, such as H2, has 1.2044 x 1024 atoms because every molecule (and therefore particle) has two component atoms.
tibertutor.com
Next Up
You have completed the sub-topic S1.4 notes, covering "Moles" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic S1.4 resources