IB Chemistry R3.3 Notes
This page contains our IB Chemistry notes for R3.3. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Electron sharing'.
Structure of alkanes
The third reaction type you need to know about is electron sharing, which typically takes the form of free radical substitution. This occurs in alkanes because they have no functional group and consist purely of C-C and C-H bonds. As a result, their general formula is CnH2n+2.
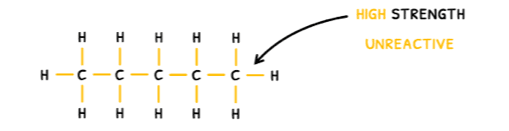
Due to their structure, alkanes are non-polar so they only have weak London dispersion forces, making them volatile and insoluble in water. However, the high strength of the C-C and C-H bonds makes them unreactive.
Thus, the only reactions they undergo are with very reactive species called radicals. Thus, during free radical substitution, alkanes reacts with halogens in the presence of UV light to form a halogenoalkane and hydrogen halide. For example:
C2H6 + Cl2 → C2H5Cl + HCl
tibertutor.com
Next Up
You have completed the sub-topic R3.3 notes, covering "Electron sharing" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic R3.3 resources