IB Chemistry R3.1 Notes
This page contains our IB Chemistry notes for R3.1. By reading each one of these notes, you will fully cover the content for IB Chemistry 'Proton transfer'.
Chapters
Ionic Theory
Topic R3.1 focuses on proton transfer reactions, the most common of which are reactions involving acids and bases. However, their properties are rather complex and require an in-depth look to understand the full process. Acids and bases can be described by three theories:
- Ionic theory - involves the formation of protons.
- Brønsted-Lowry theory - involves the transfer of protons.
- Lewis theory - involves the transfer of electrons, so covered in Topic R3.4.
Let's start with ionic theory, which is very simplistic. It has two key principles:
- Acids dissolve in water to produce H+ at a concentration greater than 1.0 x 10-7 mol dm-3.
- Bases dissolve in water and can neutralize acids.
Brønsted-Lowry theory is more complicated. This also has two principles:
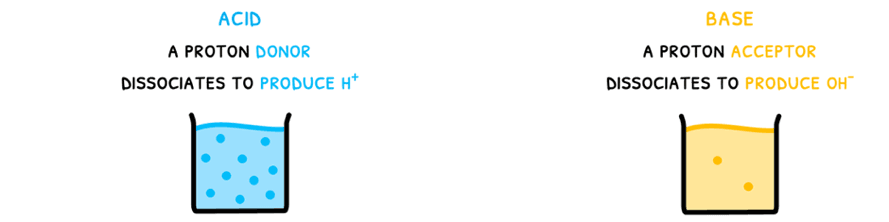
- Acids dissociate to produce H+, which is subsequently donated to another molecule.
- Bases dissociate to produce OH-, and subsequently accepts a proton.
tibertutor.com
Next Up
You have completed the sub-topic R3.1 notes, covering "Proton transfer" for IB Chemistry - continue with related resources below or explore the full IB Chemistry course from the IBO.
Other Sub-topic R3.1 resources