IB Physics Sub-topic E2 Notes
This page contains our IB Physics notes for sub-topic E2. By reading each one of these notes, you will fully cover the content for IB Physics 'Quantum physics'.
Chapters
Loading progress...
The photoelectric effect
Electromagnetic radiation is traditionally thought of as a wave. However, Einstein proposed their nature to be particular in his explanation of the photoelectric effect, the emission of electrons from a metal surface when UV light shines on it.
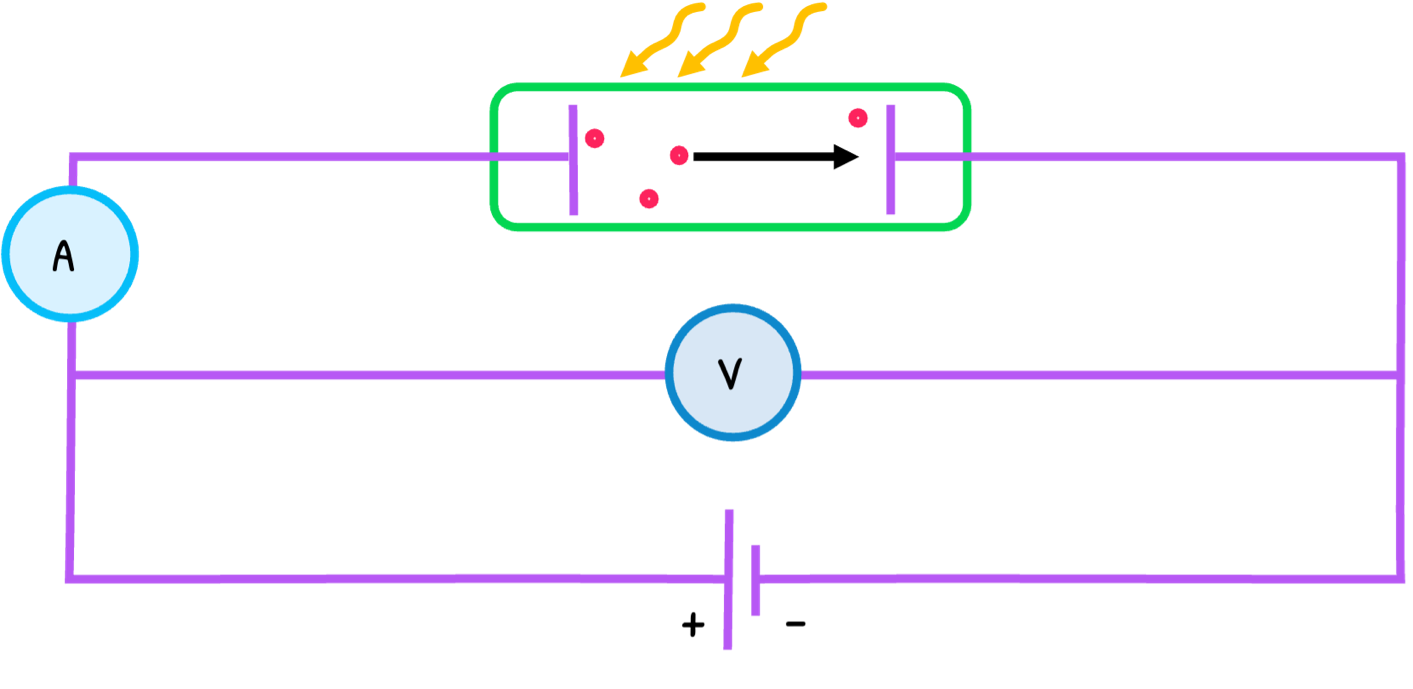
The process is as follows:
- UV light with frequency f arrives in particles called photons. Their energy E is:
- The minimum energy that electrons need to escape the energy is called the work function (Φ). Every electron absorbs a photon with a different frequency/energy and if the photon is below a threshold frequency (f0), no electron is emitted.
- If the energy is higher than the work function, the electron escapes and the remaining energy is converted into kinetic energy. The maximum kinetic energy (Emax) is:
- The number of electrons emitted depends on the UV light intensity.
- The process with a reverse supply is known as the stopping potential experiment. In this, the voltage is increased until electrons are decelerated to a point of no emission, called the stopping potential (Vs) and proportional to the electron’s maximum kinetic energy. The equation is:
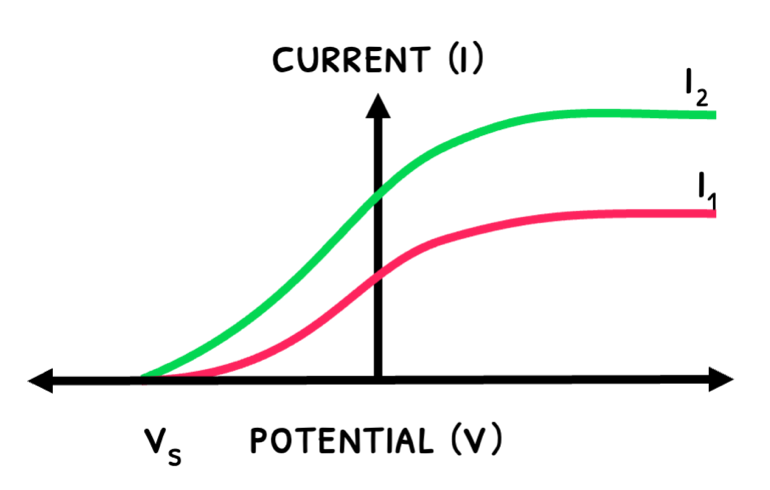
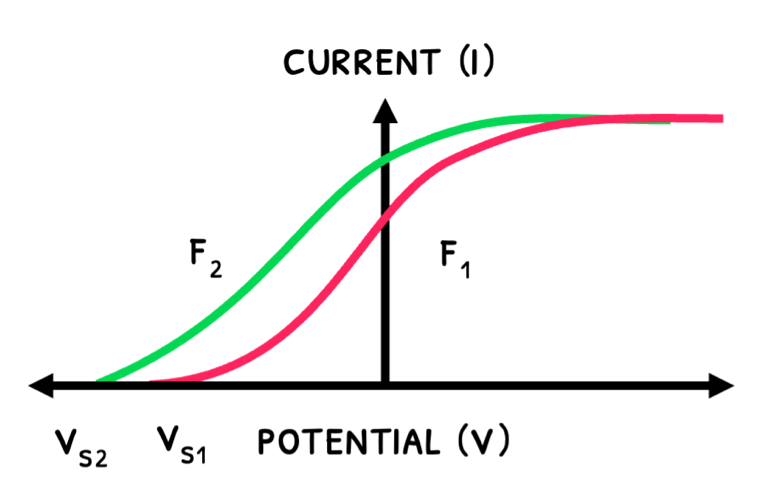
The graphs below show how the current varies with UV light’s intensity and frequency:
- An increase an intensity releases more electrons, increasing maximum current.
- An increase in frequency increases kinetic energy and stopping potential.
tibertutor.com
Great Work!
You have now covered all of our sub-topic E2 notes, covering "Quantum physics" for IB physics.
Now that you have completed these IB physics sub-topic E2 notes, covering "Quantum physics", dive into our sub-topic E2 flashcards for the IB physics course.
IB physics sub-topic E2 flashcards
Explored IB Physics?
Get stuck into one of our other subjects!
Join 85,000 students, across 130+ countries, in 500+ IB schools. That's half of the IB science graduates worldwide.
Start a 7d free trial