IB Biology A1.1 Notes
This page contains our IB Biology notes for A1.1. By reading each one of these notes, you will fully cover the content for IB Biology 'Water'.
Chapters
Structure of water
Water is the most essential molecule needed to sustain life on Earth. Also known as dihydrogen monoxide, it is composed of two hydrogen atoms covalently bonded to an oxygen atom. The sharing of electrons in these bonds is unequal, which means that water is a polar molecule!
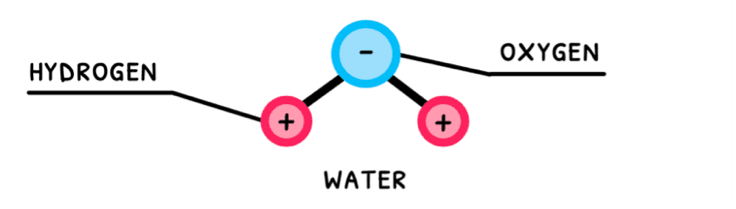
Polar molecules with hydrogen and one of nitrogen, oxygen, or fluorine exhibit hydrogen bonding. Water fits this description, and thus exhibits hydrogen bonding between its molecules. Water’s polarity and its hydrogen bonding are the reason it is so versatile and the primary molecule we search for when attempting to detect alien life.
tibertutor.com
Next Up
You have completed the sub-topic A1.1 notes, covering "Water" for IB Biology - continue with related resources below or explore the full IB Biology course from the IBO.
Other Sub-topic A1.1 resources