IB Biology B2.1 Notes
Hydrophilicity and hydrophobicity
You have previously learned that all cells contain a plasma membrane, which controls the transport of materials in and out of the cell. The structure as we know it today was formed from a series of models. However, before this is covered, it is important to understand a few key concepts: hydrophobic and hydrophilic molecules.
- Hydrophobic molecules are repelled by water and so only bond with other hydrophobic molecules. Lipids (fats) are an example of a hydrophobic molecule.
- Hydrophilic molecules are attracted to water and so only bond with water or other hydrophilic molecules. Proteins are an example of a hydrophilic molecule.
- Amphipathic molecules are partly hydrophobic and partly hydrophilic, allowing them to bond with water/hydrophobic molecules and hydrophobic molecules at the same time.
An example of an amphipathic molecule is a phospholipid, a type of fat that is a central component of the plasma membrane. Remember that phospholipids have a hydrophilic phosphate head and two hydrophobic lipid tails. As a result, lipids in the plasma membrane form a continuous sheet-like bilayer in water-based environments:
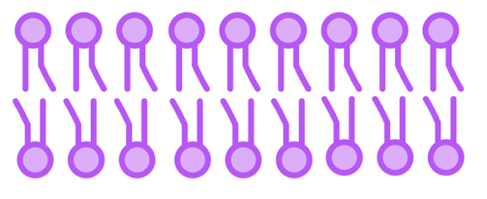
- Hydrophilic phosphate heads face outwards towards water.
- Hydrophobic lipid tails face inwards towards each other to stabilize the bilayer.
- This structure means that the membrane core has a low permeability to large molecules and hydrophilic particles, including ions and polar molecules.
Overall, having a lipid bilayer is thus an effective barrier between aqueous solutions and capable of controlling transport in and out of hte cell.