IB Biology D2.3 Notes
This page contains our IB Biology notes for D2.3. By reading each one of these notes, you will fully cover the content for IB Biology 'Water potential'.
Osmolarity
In Topic A1.1, you learned about the properties of water and that it dissolves polar substances via hydrogen bonds or dipole-dipole forces. B2.1, you learned about osmosis and aquaporins. Here, you need to learn about a key practical in biology involving potato cores in different solutions of water and observing the effects of osmosis. However, to effectively understand the practical, a few terms need to be defined first.
- Osmoles – this is the moles of solute particles that are dissolved in a solution.
- Osmolarity – this is a solution’s concentration expressed as the osmoles per liter. Sometimes you may see the term Osmolality, which is osmoles per kilogram, so remember the distinction!
With these definitions, two solutions can be compared using three terms:
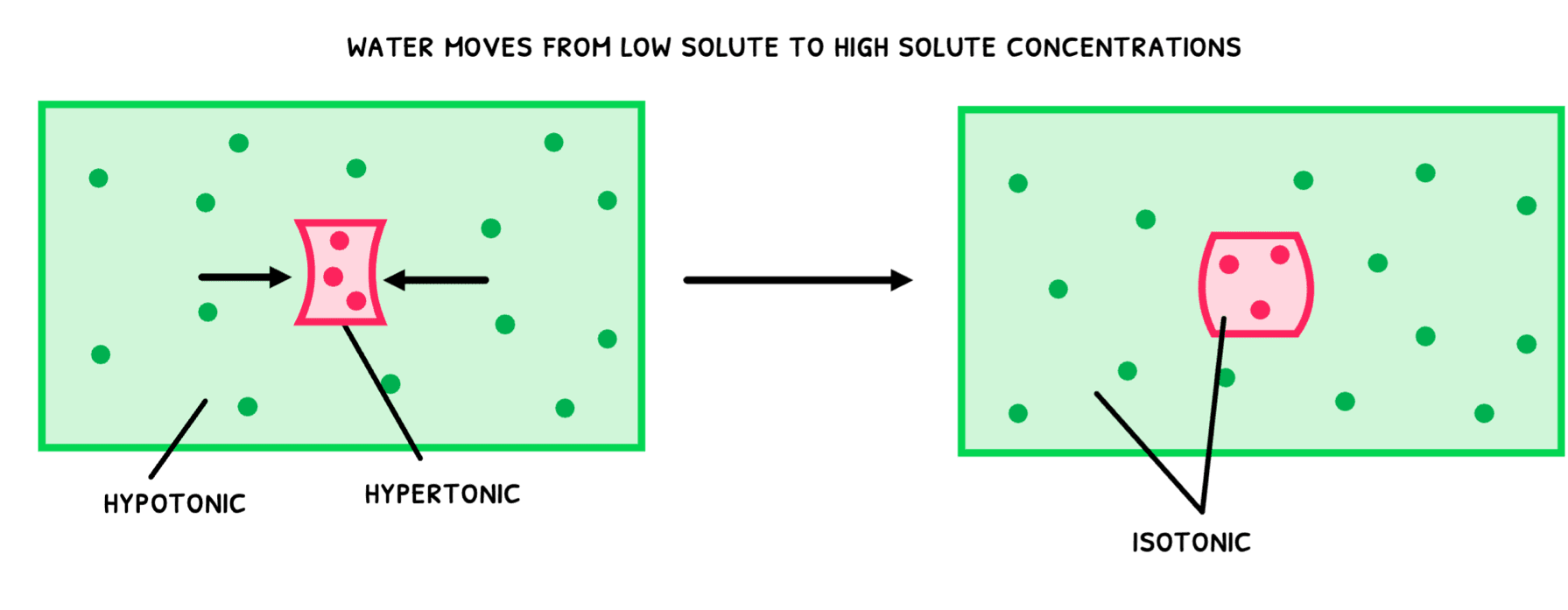
- Hypertonic – this refers to a solution with a higher osmolarity than another solution.
- Hypotonic – this refers to a solution with a lower osmolarity than another solution.
- Isotonic – this refers to two solutions with equal osmolarity.
tibertutor.com
Next Up
You have completed the sub-topic D2.3 notes, covering "Water potential" for IB Biology - continue with related resources below or explore the full IB Biology course from the IBO.
Other Sub-topic D2.3 resources